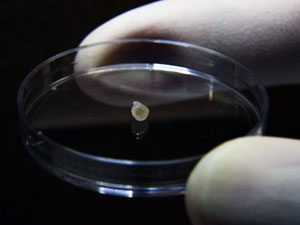
One of the tiny midbrains produced by Huck Hui Ng’s team.
© 2017 A*STAR Genome Institute of Singapore
Huck Hui Ng and his team were observing a plate of mini brains when they noticed something that made them think of the renowned French anatomist Felix Vicq d’Azyr. In 1786, D’Azyr published a treatise of the human brain, in which he described for the first time the presence of “dark spots” buried deep in the brainstem, the brain’s control center.
Huck Hui’s team was seeing the same cloudy speckles materialize in their nursery of tiny midbrains1. Since D’Azyr’s time, neurologists had discovered that the dark pigments, called neuromelanin, were produced by dopamine-secreting neurons, and that damage to these neurons causes Parkinson’s disease.
“We were very excited,” says Huck Hui at the A*STAR Genome Institute of Singapore. “It was the first time that neuromelanin had been detected in vitro.” The mottled bulbs, derived from stem cells, signaled to Huck Hui and his collaborators, Hyunsoo Shawn Je at Duke–NUS Medical School, and Eng King Tan at Singapore’s National Neuroscience Institute that they were on course to determine the biological basis for Parkinson’s. “It marked the start of a very exciting journey in trying to understand Parkinson’s disease.” The discovery also fueled increasing confidence in the use of stem cells to study complex diseases in the human brain.
Neurodegenerative diseases such as Alzheimer’s and Parkinson’s are characterized by a progressive deterioration of the nervous system. These diseases can cause declines in mental capacity, a condition known as dementia, as well as reduced control over movement, resulting in the characteristic Parkinson’s tremors. Up to 10 million people globally have Parkinson’s disease and more than 45 million suffer from Alzheimer’s or some other form of dementia. With 8 million cases of dementia diagnosed every year, those numbers are expected to triple by 2050. And the cost of caring for these patients is no small burden — estimated at US$815 billion in 2015 alone. “We hope our work will have an impact on the lives of patients afflicted with these diseases,” says Huck Hui.
Black stuff

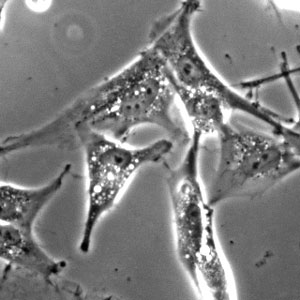
Dopamine-secreting neurons in mini models of the midbrain produce the dark pigment neuromelanin.
© 2017 A*STAR Genome Institute of Singapore
Living models of human tissue made from stem cells try to copy what our bodies naturally do. For years, researchers rushed to create model cell lines for every occasion — neurons, heart muscle cells, red blood cells. These human cell cultures, they believed, could better mimic the progression of disease in the human body than any other organism, especially the popular laboratory mouse.
But, cells in the body do not exist in segregated communities; they mix together to form complex organs. A new approach to stem cell derived models emerged around 2010, which tried instead to grow whole organs on three-dimensional (3D) scaffolds. Scientists produced rudimentary intestines, eyes and pituitary glands, but the brain remained difficult to recreate. That is, until 2013, when a group of researchers in Europe contoured its outer folds. The publication inspired Huck Hui and his collaborators in Singapore to attempt a similar model for the midbrain — a critical region in the pathogenesis of Parkinson’s disease.
They started with a batch of human embryonic stem cells, feeding and coaxing them into shape over several months. “We had to guide the differentiation in a very precise way, telling the embryonic cells what to do at every stage. It is a pretty lengthy and painful protocol,” says Huck Hui.
The researchers made their first real breakthrough in 2014: the cells formed tissue-like structures, two millimeters wide, resembling the midbrain. “We were excited, but the excitement was followed by a period of skepticism,” says Huck Hui. It wasn’t enough just to have a blob of cells sitting in the laboratory, they needed to determine whether it was brain-like in function.
They found that the neurons were definitely living, firing electric pulses back and forth when stimulated. And the neurons weren’t just any samples — they produced dopamine. The real surprise came when the two-month-old miniature organs, or organoids, formed dark deposits of neuromelanin. “This proved to us that our organ system was very different from what others had done in the past,” says Huck Hui.
The team is now trying to create mutant versions of the organoids that replicate what happens in the motor systems of patients with Parkinson’s, which is characterized by a dramatic loss of dopamine-secreting neurons.
In a clump
Similar work on stem cell models of the diseased brain is already underway in other A*STAR labs. Shi-Yan Ng at the A*STAR Institute of Molecular Cell Biology (IMCB) is using stem cells derived from patients with neurodegenerative diseases to develop 3D models of the brain. “Mini brains offer better models of aging diseases because they can mimic what is happening in a 60 year-old’s brain, unlike the fetal neurons represented by 2D cultures,” says Shi-Yan, who is looking for a very specific change which kills neural cells in the diseased brain.
Sick neurons don’t die without a sign. Their demise is precipitated by a gradual toxic accumulation of proteins: clusters of alpha-synuclein in Parkinson’s, clumps of amyloid-beta in Alzheimer’s, and a combination of many different proteins in amyotrophic lateral sclerosis (ALS).
Researchers believe that these lethal clusters can spread like an infectious plague between neurons, and even to supportive glial cells. Shi-Yan’s models could help to explain if and how these toxins seep through brain tissue. The results could have implications for surgery because sterilizing instruments does not eliminate the proteins. Depending on how the proteins are found to aggregate, surgeons may be guided to destroy their instruments after operating on patients with Parkinson’s disease.
These are early days for Shi-Yan, but so far, she has been able to create spherical brain organoids of Parkinson’s disease, in which the neurons die at a faster rate than in healthy organoids. More intriguing, however, is that the diseased cells accumulate alpha-synuclein proteins in dense clusters — something that has never been seen before in a cell model.
The methods developed by Shi-Yan could also be used to predict whether a healthy young adult test subject will develop Parkinson’s disease in 50 years. Researchers could take samples of their cells, age them by a few decades, and watch for any pathogenic effects. “We’d be able to see neurons dying within a month, which is really quick,” she says.
In the blood vessels
Brain-specific vascular smooth muscle cells.
Reproduced from Ref. 2 and licensed under CC BY 3.0 © 2014 C. Cheung et al.
Researchers could detect neurodegeneration even earlier if they looked beyond the nervous system. Christine Cheung at the IMCB has taken a hint from the famous words of 17th century English physician, Thomas Sydenham: “A man is as old as his arteries.”
“Blood vessels are like silent killers,” says Christine. This is especially apparent in a form of cognitive decline caused by blocking the brain of its blood supply, such as during a stroke. “In vascular dementia, blood vessel damage occurs before the neurons begin to die,” says Christine. “With the current focus on preventative medicine, there is cause for looking at early events.” Christine is growing stem cell models of blood vessels to better understand how one system affects the other.

Blood vessel cells are capable of clearing amyloid-beta proteins (red), which are transported to organelles (blue) for enzymatic degradation.
Reproduced from Ref. 2 and licensed under CC BY 3.0 © 2014 C. Cheung et al.
Not all blood vessels are the same, however. Unlike blood vessels to the other organs, those to the brain have to meet a higher demand for nourishment to feed our thoughts, memories and coordinated movements. The vessels are structurally distinct, and are believed to play a role in draining the brain of the toxic amyloid-beta proteins found in Alzheimer’s patients. But these functions are not visible using standard models of blood vessels.
In 2014, Christine grew the first brain-specific line of blood vessels2. She derived them from a specific type of embryonic tissue known as the neural crest, made with stem cells. The blood vessel cells clearly showed that the brain’s vasculature is essential for clearing out excess amyloid-beta proteins. Vessels exposed to low-oxygen conditions, typical in a stroke, could not pump the proteins out as efficiently. “Without organ-specific blood vessel cells to model disease processes, it is hard to have an accurate picture of what could have gone wrong.”
Christine sees huge potential for introducing her vascular cells to the 3D organoids developed by Huck Hui and Shi-Yan for tissue engineering applications.
“We are all taking small steps toward the long-term goal of coming up with more effective drugs for neurodegenerative diseases,” says Shi-Yan. “In a few years, someone will piece all these disparate studies together like a jigsaw puzzle and finally solve it.”
The A*STAR-affiliated researchers contributing to this research are from the Genome Institute of Singapore and the Institute of Molecular and Cell Biology.




