Because of its high sensitivity and accuracy, quantitative RT-PCR is currently the gold standard for diagnosing COVID-19 infection in the acute phase of the illness. However, there are certain limitations: apart from being costly and time-consuming, false negative results may arise due to insufficient viral genetic material, biological variation or improper handling of nucleic acid samples.
This has motivated industry and academic scientists to consider immunoassays, a rapid test format that may deliver results in a shorter time window and at lower cost. To date, tests are commercially available and can be used to detect SARS-CoV-2 antibodies from patient serum or plasma samples.
In this review, my colleagues and I at the A*STAR Singapore Immunology Network (SIgN) and the National University of Singapore provide an overview of the immunoassays currently available for SARS-CoV-2 detection. We also discuss the strengths, limitations and applications of this diagnostic technique, and what to expect next.
We wrote this review to help clinicians understand serology assays better, building on our experience working on serology for infectious diseases such as Zika and chikungunya. We also clear up any misconceptions about serology testing and clarify the technical parts of it without making it too complicated for non-immunologists.
Which antibodies arise and when?
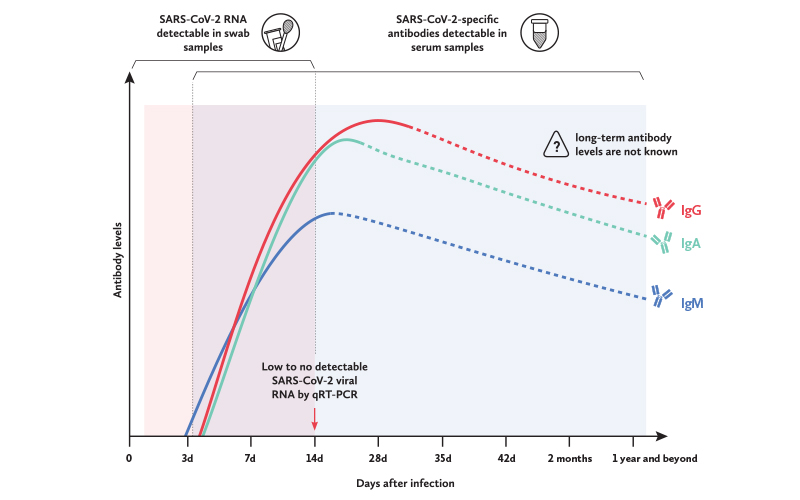
To draw reliable conclusions from immunoassays, it is important to note that the timing of testing matters greatly, since it provides a snapshot of an individual’s antibody profile at that point in time.
According to recent studies, both anti-SARS-CoV-2 IgM and IgG antibody levels increase gradually along with infection phases. IgM was detected as early as three days post-illness onset, peaking between two to three weeks post-illness onset, while IgG was detected as early as four days post-illness onset, peaking after 17 days post-illness onset.
Similar to SARS-CoV and the Middle East respiratory syndrome coronavirus (MERS-CoV), both IgM and IgG levels in SARS-CoV-2 patients appear to be correlated with disease severity, with a higher level of both antibodies present in patients with more severe infections.
However, one study showed no specific chronological order in terms of IgM and IgG seroconversion. In addition, there also seemed to be no correlation between seroconversion rates and age, gender or time of hospitalization. Thus, these findings suggest that it may be important to test for both IgM and IgG antibodies to confirm a positive infection.
Identifying antibody targets
Current serology assays mainly target two immunogenic coronavirus proteins: S protein, which is the most exposed viral protein; and N protein, which is abundantly expressed during infection. The receptor-binding domain (RBD) of the S protein is also a target of interest.
To reduce the time needed to develop novel neutralizing antibodies, it may be possible to use computational prediction to identify existing SARS-CoV antibodies that may recognize SARS-CoV-2 epitopes, the specific part of the target protein that binds to the antibody. For example, 49 out of 298 linear SARS-CoV-derived B-cell epitopes have an identical match with SARS-CoV-2 protein sequences, with the majority of matches located at the S and N proteins. Further mapping revealed several regions on the S2 subunit that may allow cross-neutralization of both SARS-CoV and SARS-CoV-2.
Beyond the development of new immunoassays, studying SARS-CoV-2 epitopes where SARS-CoV-2-specific antibodies bind to may also help guide the design of a vaccine.
© A*STAR Research
Ensuring tests are specific to SARS-CoV-2
As the majority of human populations have had prior exposure to endemic human coronavirus infections, it is necessary to validate the specificity and sensitivity of current immunoassays against SARS-CoV-2 to avoid false positive outcomes.
The high level of cross-reactivity between SARS-CoV and SARS-CoV-2 can be attributed to the high degree of genetic homology. Detailed analysis reveals a highly conserved S2 subunit domain across the coronaviruses. Thus, using an S1 subunit-based immunoassay may be more specific than the entire S antigen for diagnosing SARS-CoV-2 infections.
Since respiratory diseases are the hallmark of coronavirus infections, several studies have exploited the detection of IgA antibodies to diagnose SARS-CoV-2 infections. However, IgA-based immunoassays have been hypothesized to be less specific than IgG-based ELISA assays due to cross-reactivity with serum samples from patients infected by other coronaviruses.
Given the availability of immunoassays against various coronavirus structural proteins, it may be prudent to use more than one antigen-based serological approach to establish a true positive SARS-CoV-2 infection. In addition, the use of saliva samples and other bodily fluid swabs could also be explored as a less invasive alternative for sample collection.
Limitations of serology testing
While it may be fast, robust and easy to perform, one of the major setbacks of serology testing is its inability to detect the presence of infection during the early stages of disease, as antibodies take several days to weeks to be generated after exposure to foreign material. As such, serological testing may provide false negative results for an early SARS-CoV-2 infection and the use of quantitative RT-PCR may be more suitable in acute cases.
Furthermore, due to the unique genetic makeup of each individual, an inherent variability in antibody response could explain the variety of antibody profiles elicited among COVID-19 patients.
Cross-reactivity could be another limitation of immunoassays as it severely impacts the specificity and sensitivity of the test. Although the phylogenetically closest coronavirus, SARS-CoV, has not been reported to be circulating in the human population since 2004, other endemic human coronaviruses may still pose a problem to the accurate diagnosis of a SARS-CoV-2 infection.
Potential for surveillance, detection and intervention
Given the rapid rise in the number of confirmed COVID-19 cases coupled with the shortage of test kits, decentralized point-of-care tests may provide a cheap and straightforward option for SARS-CoV-2 diagnosis. The lateral flow assay, for example, is a paper-based platform used to detect and quantify analytes in complex mixtures.
Ultimately, the goal of serology testing for SARS-CoV-2 is to achieve an axis of surveillance, detection and intervention. Surveillance is how can you anticipate or recognize signs of an upcoming outbreak, while detection is about developing tools to identify the new pathogen. Last but not least, intervention covers measures to prevent the virus from spreading, such as by using face masks and social distancing, and ultimately vaccines.
It is also for these reasons that the A*STAR Infectious Diseases Horizontal Technology Center (ID HTC), where I serve as Executive Director, was established. We will always have infectious disease because Singapore is based on open economy requiring goods exchange and travel. Once country borders are re-opened, and as soon as you facilitate the movement of people, goods and animals, you facilitate the movement of pathogens.
The idea of the Horizontal Technology Center is to bring together different pockets of expertise on a regular basis. We hope to consolidate what we have learnt about COVID-19 to help us prepare for future outbreaks.
The A*STAR-affiliated researchers contributing to this research are from the Singapore Immunology Network (SIgN) and the Infectious Diseases Horizontal Technology Center (ID HTC).




