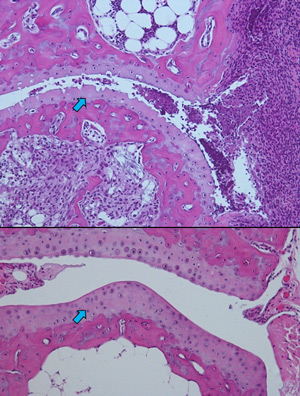
Joint tissue sections show that relative to untreated animals (top), treatment with the P2D7 antibody (bottom) greatly reduces cartilage loss (blue arrows).
Reproduced from Ref. 1 and licensed under CC BY-NC 3.0 © 2014 A. X. H. Goh et al.
Through a combination of natural immune selection and targeted protein engineering, A*STAR researchers have generated an antibody that efficiently blocks a disease-related inflammatory pathway in both human cells and mouse experimental models.
The signaling protein interleukin-1β (IL-1β) helps marshal a rapid immune counterattack in the event of infection or injury. However, excess IL-1β activity can result in damaging inflammation, contributing to disease states ranging from gout and diabetes to various autoimmune disorders. IL-1β inhibitors are an active area of drug discovery, and Cheng-I Wang and colleagues at the Singapore Immunology Network recently set out to generate an IL-1β-specific antibody that might prove useful for tackling inflammatory disease.
The mammalian immune system has evolved to generate antibodies that can bind to foreign molecules with remarkable affinity and specificity: scientists have streamlined this by developing experimental methods that offer a simpler means for obtaining such antibodies. Wang and colleagues took such an approach, using protein engineering to obtain a promising antibody that proved capable of binding and inhibiting both human and mouse IL-1β.
The researchers focused on a small number of amino acids in the antibody that were likely to contribute to binding, and generated a library of antibody variants with random substitutions at these sites. By screening this library, Wang and colleagues obtained antibodies with remarkably improved performance relative to the starting molecule. “We were surprised to see the extent of the improvement,” he says. “We obtained clones that showed 20- to 50-fold better affinity.”
The best of these antibody variants, P2D7, proved 11 times more effective at inhibiting IL-1β than canakinumab, a commercially available anti-inflammatory drug. Canakinumab and P2D7 bind the same protein but recognize distinct targets on that molecule. Furthermore, P2D7 binds to the mouse and monkey versions of the IL-1β protein with high affinity, whereas canakinumab does not. “This will make it possible for us to use this antibody in various in vivo disease models,” says Wang. As a proof of concept, the researchers used mouse models to show that P2D7 could counter the symptoms of arthritis (see image) and peritonitis, or even increase survival in animals injected with human myeloma cells.
This demonstrates the clear value of P2D7 as a research tool, but Wang’s team is also exploring the clinical potential of this antibody. “Together with a pharmaceutical company, we are planning to study the applications of P2D7 in disease areas that are less explored, in which IL-1β may also play a role,” he says.
The A*STAR-affiliated researchers contributing to this research are from the Singapore Immunology Network.




