As part of the body’s frontline defense against microbial enemies, neutralizing antibodies have been thrust in the spotlight for their potential to treat COVID-19. Now, research led by Cheng-I Wang, a Principal Investigator at A*STAR’s Singapore Immunology Network (SIgN), has uncovered a darker side to some of these immune heroes.
Previously, Wang and colleagues demonstrated that most neutralizing antibodies block the SARS-CoV-2 spike protein’s receptor-binding domain (RBD) from interacting with ACE2 receptors on host cells. However, the influence of these antibodies on the generation of syncytia—cell fusions associated with severe COVID-19 symptoms—has remained a gray area.
“The formation of syncytia has been linked to lung tissue damage observed in severe COVID-19 cases when cell-cell fusion occurs inside the patient’s airways,” explained Wang, adding that clearer guidelines for selecting neutralizing antibodies as treatments for COVID-19 patients with severe lung damage are urgently needed.
In a study published in Cell, Wang teamed up with collaborators to analyze a panel of six antibodies for factors that may impact SARS-CoV-2 neutralization potency and syncytia formation. These include properties such as how strongly the antibody binds to its target and where the antibody binds to the spike protein.
As predicted, all six antibodies blocked interactions between the SARS-CoV-2 spike protein and ACE2 receptors, but unexpectedly displayed a range of responses to syncytia formation.
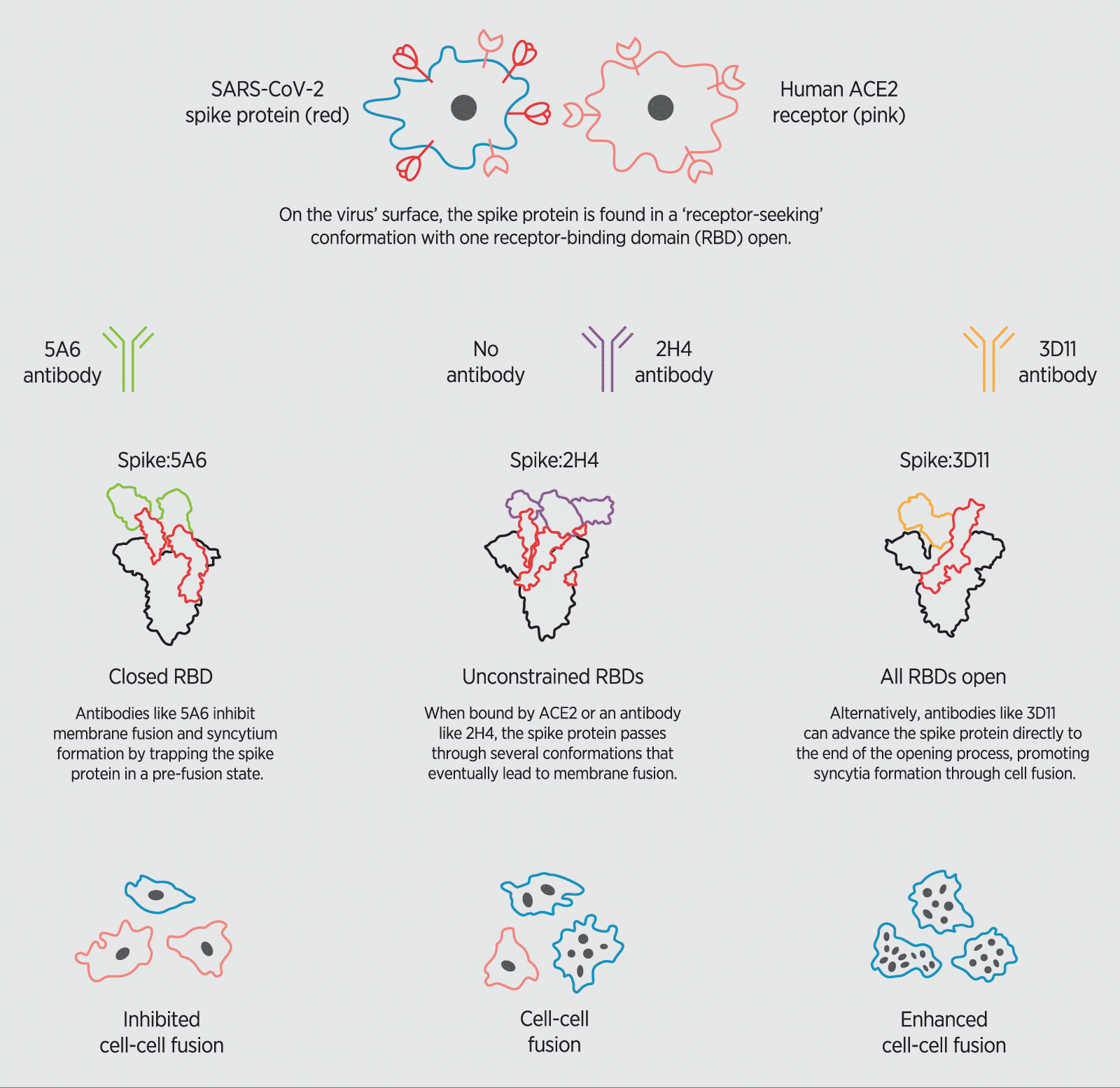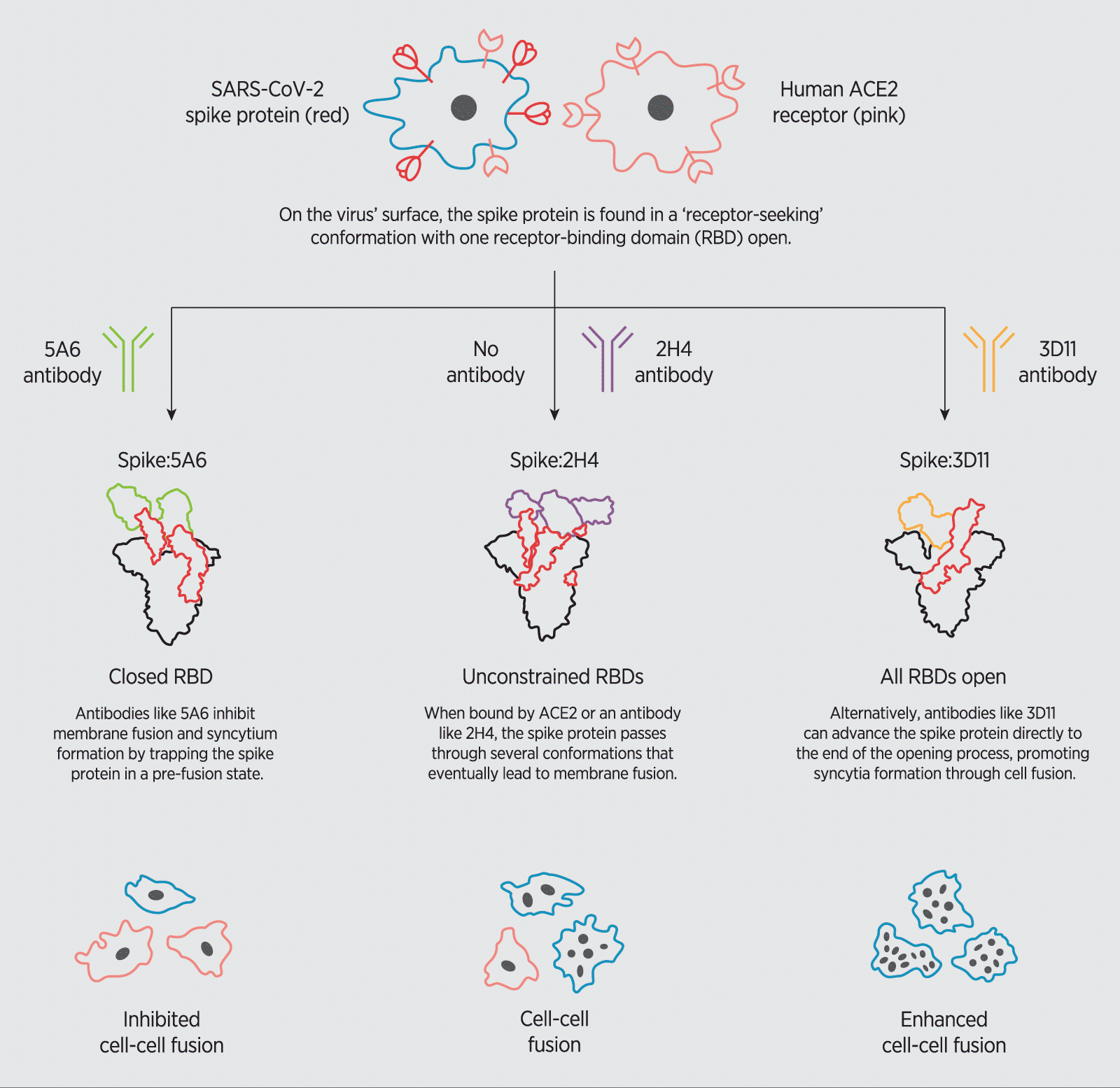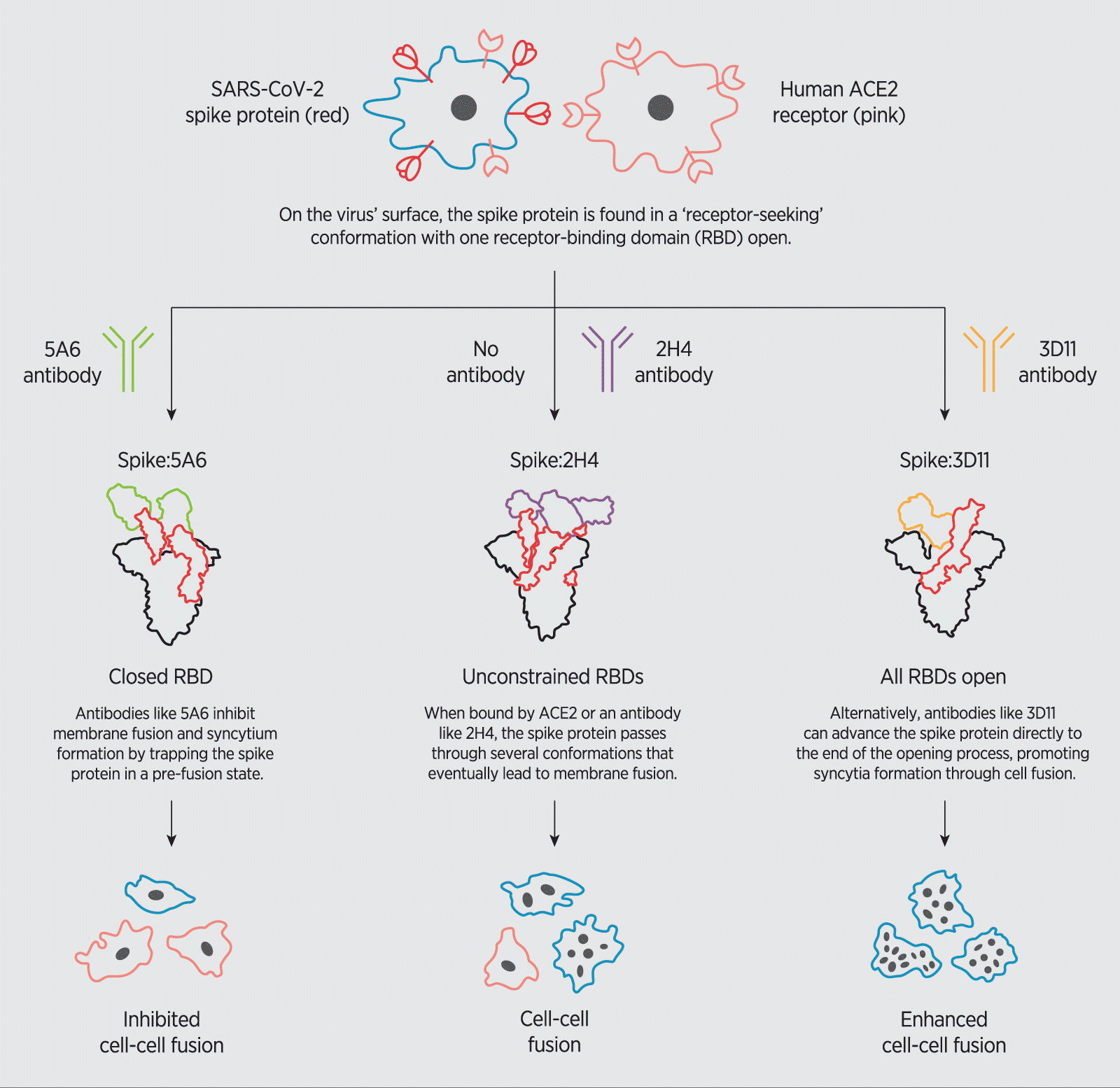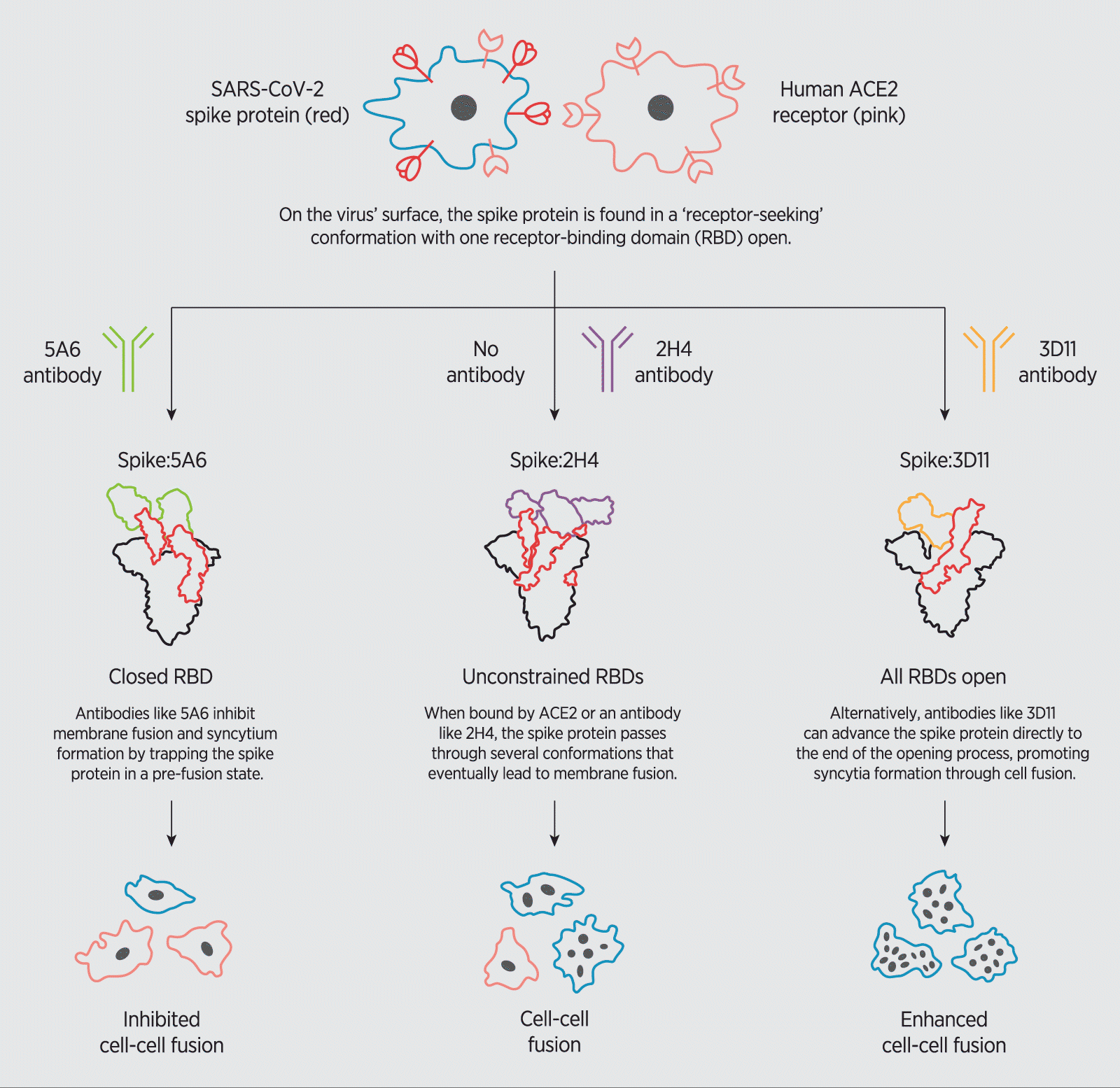
How SARS-CoV-2 neutralizing antibodies modulate the formation of syncytia.
© A*STAR Research
An antibody called 3D11, for instance, displayed the strongest binding to the spike protein’s RBD—which by current standards, would suggest potential therapeutic value. Interestingly, however, 3D11 attaches to the RBD at a site that locks the spike protein in an ‘open’ pre-binding position, effectively accelerating interactions between the spike protein and ACE2 as well as promoting syncytia formation.
Conversely, the 5A6 antibody bound less strongly to the spike protein, but was far more potent, latching onto the RBD and strongly inhibiting syncytia development. A closer look at the structural dynamics of 5A6 binding to the RBD revealed that the antibody recognized an exposed loop near the tip of the RBD, trapping the spike protein in its ‘closed’ position, thereby hindering ACE2 binding.
“These findings suggest that the potential effectiveness of a neutralizing antibody is influenced by several possibly countervailing factors, highlighting a complex basis for viral neutralization potency,” Wang concluded. The team is now looking to partner with industry collaborators to commercialize the 5A6 antibody as a viable candidate for COVID-19 treatment.
The A*STAR-affiliated researchers contributing to this research are from the Singapore Immunology Network (SIgN).




