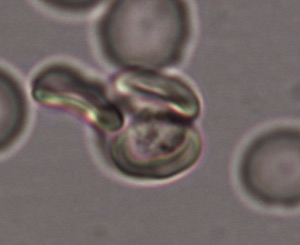
Malaria-infected red blood cells bind to uninfected red blood cells, forming structures called ‘rosettes’.
© 2014 A*STAR Singapore Immunology Network
The distinctive ‘clumping’ of blood cells that blocks vessels and causes tissue damage in malaria-infected patients is the focus of a multinational collaboration, which includes A*STAR researchers.
When malaria parasites are released into a host following a mosquito bite, they make their way into red blood cells (RBCs). In some cases, infected RBCs stick to uninfected RBCs — a phenomenon known as ‘rosetting’ (see image) leading to the formation of structures called rosettes. Studies of Plasmodium falciparum, the species responsible for the most malaria cases worldwide, suggest that rosetting either assists with the infection of new cells or acts as a shield against immune detection. The process can also contribute to certain complications associated with malaria.
Researchers led by Bruce Russell of the National University of Singapore recently set out to establish the function of rosetting in Plasmodium vivax, a less-well-understood species that nevertheless is responsible for almost 350 million cases of malaria per year. Laurent Renia, a collaborator on the study from the A*STAR Singapore Immunology Network, notes that the study of P. vivax relative to P. falciparum has been limited by the lack of robust experimental culture models. “P. vivax grows exclusively in reticulocytes, the young form of RBCs, which represent only 1 to 2 per cent of circulating RBCs in a normal person,” he says. “In contrast, P. falciparum grows in normocytes, which are mature RBCs.”
The researchers employed recently developed techniques for working with patient blood samples to analyze rosetting in P.-vivax-infected RBCs. They determined that cells infected by P. vivax and P. falciparum form rosettes with similar frequencies. Surprisingly, most P. vivax rosettes incorporated normocytes rather than reticulocytes, even though the latter cells are the preferred target of this species. “This mechanism was thought to facilitate the invasion of uninfected RBCs by reducing the distance between the parasite and the target cell,” says Renia. “Instead, rosetting could help the parasite to escape the host immune system.”
The researchers also learned details about how rosettes form. By using antibodies to block specific proteins on the surface of RBCs, they determined that a molecule called glycophorin C contributes to P.-vivax-mediated rosetting. Indeed, rosetting was rarely detected with RBCs lacking glycophorin C.
The other side of the puzzle remains unclear, however — the P. falciparum protein responsible for rosetting is absent in P. vivax, and Renia is now keen to uncover this missing molecule. “If we identify the parasite protein at the surface of the red blood cells, we can try to develop a compound to block this interaction,” he says.
The A*STAR-affiliated researchers contributing to this research are from the Singapore Immunology Network.




