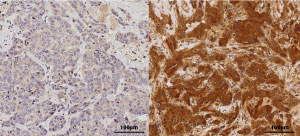
Primary (left) and recurrent (right) tumors images stained with p-IRAK1.
© 2016 A*STAR Genome Institute of Singapore
Triple negative breast cancer (TNBC), a particularly aggressive form of the disease, has one of the highest rates of recurrence. A*STAR researchers have now shown that by blocking the activity of a particular enzyme, targeted therapy for metastatic TNBC may finally become a reality.
A lack of effective, targeted therapy motivated Yu Qiang from the A*STAR Genome Institute of Singapore and colleagues to take a closer look at the pathways involved in TNBC metastasis and chemoresistance.
About 15 to 20 per cent of all breast cancers are ‘triple negative’, which means they lack estrogen or progesterone receptors that can be targeted by hormone therapy, as well as the protein HER2 which may be targeted directly by drugs such as Herceptin. This leaves chemotherapy drugs as among the few options available for treating TNBC. These drugs do not discriminate between healthy and cancerous cells. Patients can develop a resistance to common chemotherapy drugs, such as paclitaxel during initial treatments, leaving them vulnerable to rapid disease progression in the case of a recurrence.
In exploring TNBC metastasis, the A*STAR team found that an enzyme typically associated with inflammation response — interleukin-1 receptor-associated kinase 1 (IRAK1) — was ‘over-expressed’ in TNBC. Upon treatment with paclitaxel, this IRAK1 becomes strongly phosphorylated, and is, as Yu notes “highly enriched in recurrent breast tumors, which suggests it can potentially be a useful marker for predicting tumor recurrence.”
IRAK1 was found to both drive aggressive growth and metastasis of cancerous cells through the nuclear factor-κB pathway — a signaling pathway linked to cancer progression — and strengthen paclitaxel resistance. “We realized then that IRAK1 could be a new therapeutic target for both TNBC metastasis and chemoresistance,” Yu recalls.
By administering an IRAK1 inhibitor — such as ginseng derivative, Compound K — which blocked IRAK1 activity and triggered paclitaxel-induced apoptosis, the team found that not only could the chemoresistance be reversed, the TNBC progression could also be slowed. “We could speculate that upfront administering of drugs that can block IRAK1 activity might be able to prevent tumor recurrence,” remarks Yu.
The team is working to develop more inhibitors to tackle IRAK1 at this important juncture. “Our long-term goal is to develop drugs that target IRAK1 for a novel combination approach, to efficiently trigger apoptosis in recurrent metastatic cancer cells,” says Yu. “By achieving this, we will be able to generate therapeutic effects by eliminating metastatic burden and overcome drug resistance.”
The A*STAR-affiliated researchers contributing to this research are from the Genome Institute of Singapore. For more information about the team’s research, please visit the GIS Cancer webpage.




