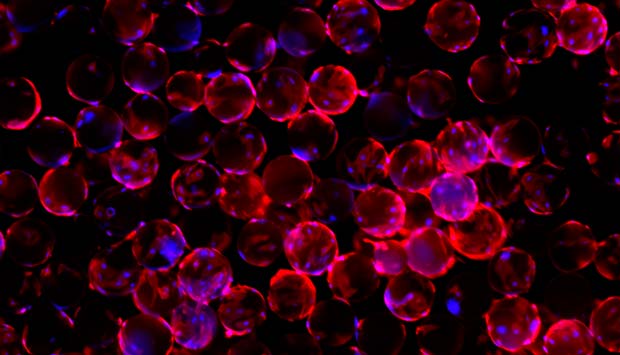
Stem cells (red) on polycaprolactone-based microcarriers.
Reprinted from Ref.1, Copyright 2016, with permission from Elsevier.
Bone tissue engineering is theoretically now possible at a large scale. A*STAR researchers have developed small biodegradable and biocompatible supports that aid stem cell differentiation and multiplication as well as bone formation in living animal models.
Mesenchymal stem cells self-renew and differentiate into fat, muscle, bone, and cartilage cells, which makes them attractive for organ repair and regeneration. These stem cells can be isolated from different sources, such as the human placenta and fatty tissue. Human early mesenchymal stem cells (heMSCs), which are derived from fetal bone marrow, were thought to be best suited for bone healing, but were not readily accessible for therapeutic use.
Existing approaches to expand stem cells for industrial applications tend to use two-dimensional materials as culture media, but their production yields are too low for clinical demand. Furthermore, stem cells typically need to be harvested with enzymes and attached to a scaffold before they can be implanted.
To bring commercially viable cell therapies to market, Asha Shekaran and Steve Oh, from the A*STAR Bioprocessing Technology Institute, have created directly implantable microscopic spheres in collaboration with the A*STAR Institute of Materials Research and Engineering. These spheres, which acted as heMSC microcarriers, consist of a biodegradable and biocompatible polymer called polycaprolactone.
According to Shekaran, their initial aim was to expand stem cells on microcarriers in bioreactors to scale up production. However, this strategy threw up difficulties, especially when attempting to effectively dissociate the cells from the microcarriers and transfer them to biodegradable scaffolds for implantation.
“A biodegradable microcarrier would have a dual purpose,” Shekaran says, noting that it could potentially provide a substrate for cell attachment during scalable expansion in bioreactors, and a porous scaffold for cell delivery during implantation.
The researchers generated their microcarriers by synthesizing polycaprolactone spheres and coating them with two proteins polylysine and fibronectin. These proteins are found in the extracellular matrix that assists cell adhesion, growth, proliferation, and differentiation in the body.

Stem Cell group members Eileen Sim and Asha Shekaran develop scalable and cost-effective technologies for stem cell expansion and bone tissue engineering.
© 2017 A*STAR Bioprocessing Technology Institute
Microcarriers that most induced cell attachment also promoted cell differentiation into bone-like matrix more strongly than conventional two-dimensional supports. In addition, implanted stem cells grown on these microcarriers produced an equivalent amount of bone to their conventionally-derived analogs.
“This is encouraging because microcarrier-based expansion and delivery are more scalable than two-dimensional culture methods,” says Shekaran.
The team now plans to further investigate the therapeutic potential of these microcarrier–stem cell assemblies in actual bone healing models.
The A*STAR-affiliated researchers contributing to this research are from the Bioprocessing Technology Institute and the Institute of Materials Research and Engineering. For more information about the team’s research, please visit the Stem Cell Group webpage.