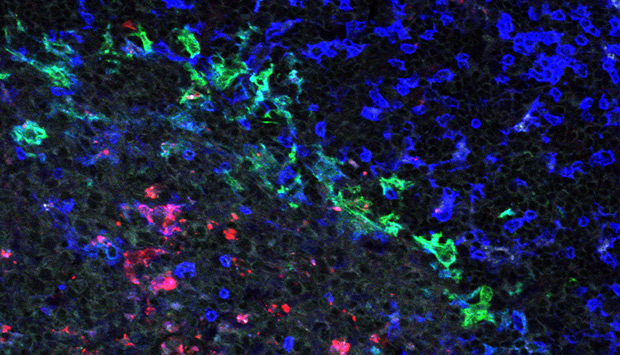
Fluorescence microscopy image of a spleen from a dengue virus-infected mouse with infected cells labeled red, macrophages labeled green and T cells labeled blue.
© 2014 A*STAR Singapore Immunology Network
Dengue virus infects hundreds of millions of people living in tropical countries every year. Transmitted via mosquito bites, the virus typically causes fever, but may also lead to potentially fatal organ failure. The development of mouse models of dengue virus infection could hasten an effective response to the disease, for which no specific treatments or licensed vaccines currently exist.
Now, an international team of researchers led by Katja Fink at the A*STAR Singapore Immunology Network has investigated small alterations in the normal mouse immune system to allow for the study of dengue virus infection, treatment and prevention.
Mice with a normal immune system are unable to be infected by dengue virus, which means that they cannot be used as model systems to understand dengue infection in humans. On the other hand, mice lacking normal interferon responses, which are generally required to fight viral infection, can be infected with the virus. But these mice do not mount a normal immune response, which does not make them very useful for learning about how the immune system reacts to dengue virus infection. Fink and colleagues reasoned that it would be highly beneficial to the field to have a mouse model that could be both infected with the virus and exhibit as normal an immune response as possible.
“Dendritic cells and macrophages are immune-cell types that had been previously shown in humans and mice to be potential targets for dengue virus infection, and both are key mediators of the immune response,” explains Fink. “Therefore, we thought ablating interferon signaling in one or the other cell type could be sufficient to make mice susceptible to infection by the dengue virus,” she says.
Fink and team found that mice lacking interferon signaling in T cells, another type of immune cell, could not be infected, whereas those lacking interferon signaling in dendritic cells or macrophages were killed by a dengue virus infection. However, the researchers further showed that mice lacking interferon signaling in either dendritic cells or macrophages could also mount an immune response to the infection, including the activation of T cells that could target the virus (see image). The immune system of these mice could also generate an effective neutralizing antibody response to a dengue vaccine candidate.
The development of these mouse models of dengue virus infection could allow for rapid progress in research into potential dengue virus treatments or vaccines that could be efficacious in humans.
The A*STAR-affiliated researchers contributing to this research are from the Singapore Immunology Network.