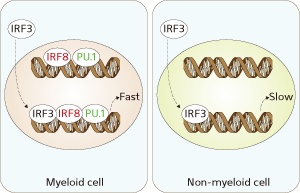
In response to pathogenic stimulation, the pre-formed activation complex of IRF8 and PU.1 facilitates rapid recruitment of activated IRF3 to the IFN-β promoter (left), thereby enabling prompt expression of IFN-β. In non-myeloid cells lacking a pre-bound IRF8 and PU.1 priming complex, IFN-β transcription is delayed (right).
Interferon-β (IFN-β) is an inflammatory factor important for mobilizing the immune system to fight invading pathogens. Keh-Chuang Chin at the A*STAR Singapore Immunology Network and co-workers have now discovered three transcription factors that cooperate to rapidly drive IFN-β expression in human immune cells called monocytes in response to pathogen exposure.
Chin and his co-workers wanted to know why monocytes were able to increase the expression of IFN-β so rapidly after being exposed to pathogens, whereas other cell types had a much slower induction of IFN-β. They thought that the transcription factor interferon regulatory factor 8 (IRF8) may be involved, because it is expressed in the myeloid family of immune cells, which include monocytes.
The researchers found that bacterial factors could increase the expression of IRF8 in human monocytes. Knocking down IRF8 blocked the induction of IFN-β in monocytes, while the expression of IRF8 in mouse myeloid progenitor cells that do not normally contain this transcription factor made the cells capable of driving IFN-β expression (see image).
The researchers demonstrated that IRF8 could bind to the IFN-β gene in monocytes even before treatment with bacterial factors. This suggested to the research team that IRF8 tended to sit on the IFN-β gene, ready at any moment to be called into action to drive IFN-β transcription in human monocytes. This could account for the ability of these cells to very rapidly induce expression of IFN-β.
IRF8 has previously been found to interact with the transcription factor PU.1. Chin and his colleagues showed that PU.1 also sits on the IFN-β gene in monocytes, even in the absence of pathogen exposure, and is required for the induction of IFN-β expression. Human kidney cells that do not contain either IRF8 or PU.1 were able to induce IFN-β only if the researchers expressed both transcription factors in the cells.
The transcription factor IRF3 can also bind to the IFN-β gene, and the researchers found that IRF3 could interact with IRF8 in human monocytes. They showed that IRF8 brings IRF3 to the IFN-β gene and allows IRF3 to aid in IFN-β transcription.
The findings identify a complex of three transcription factors that is responsible for inducing IFN-β expression in monocytes and therefore in orchestrating the body’s rapid response to invading pathogens. “The discovery of this complex may open a new way for scientists to develop strategies to target inflammatory diseases related to misregulation of the interferon response,” explains Chin.
The A*STAR-affiliated researchers contributing to this research are from the Singapore Immunology Network.



