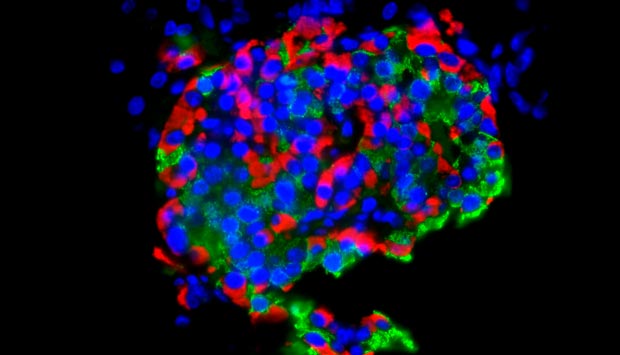
A*STAR researchers show that HNF1B mutation affects the development of the pancreas in MODY5 patients which means they develop diabetes by early adulthood. The image above depicts a human islet containing alpha and beta cells.
© 2016 A*STAR Institute of Molecular and Cell Biology
Personalized treatment for people with diabetes could be a step closer after researchers discovered how a single gene mutation fundamentally alters pancreatic development.
The A*STAR researchers discovered that a point mutation in a gene known as HNF1B halts the embryonic development of the pancreas; it stops the growth of the vital beta cells, which produce the insulin that maintains a body’s sugar balance.
Improved understanding of beta cell development could lead to better-targeted diabetes treatments, explains study leader Adrian Teo, from the A*STAR Institute of Molecular and Cell Biology (IMCB).
“By understanding the role of these beta cell genes, we can understand the mechanistic cause of diabetes,” Teo says. “This may mean that eventually we could stratify our diabetic population into subgroups and treat them appropriately based on disease mechanism rather than just blood glucose levels”.
The team used pluripotent stem cell lines from people with a rare form of inherited diabetes called maturity onset diabetes of the young, type 5 (MODY5), comparing them to cells grown from unaffected family members and healthy controls.
In MODY5, children who inherit the gene mutation from one parent grow a malformed and small pancreas, which means they develop diabetes usually before the age of 25.
“We used MODY5 patient-specific induced pluripotent stem cells to go back in time, mimicking human pancreas development in our cell culture dish,” Teo explains. “Amazingly, we discovered that HNF1B gene mutation actually affected the development of the human pancreas very early in life.”
While there are a number of subtypes of MODY associated with a range of gene mutations, the team found HNF1B worked independently of all of those. In the early days of development the differentiating cells actually up-regulated other pancreatic development genes, in what may have been an attempt to compensate for the HNF1B mutation.
“We saw the same effects in numerous affected cell lines that were clearly distinct from the healthy family member and non related healthy control,” Teo says.

Adrian Teo (back row, first from the left) and the IMCB team.
© 2016 A*STAR Institute of Molecular and Cell Biology
“Together, these comparisons allowed us to study human diabetes mechanisms in an actual genetic background which is different to other models that typically do not account for true human genetic background.”
The team are now using a similar pluripotent stem cell method to examine the pancreatic defects involved in other forms of diabetes.
“Diabetes is a complex disease arising from a combination of genes and environment, but this model can help us understand the contribution that individual genes make to beta cell function,” Teo explains.
The A*STAR-affiliated researchers contributing to this research are from the Institute of Molecular and Cell Biology.